Shares in CytoDyn lifted after the biotech startup said its new drug showed good signs as a treatment for mild-to-moderate coronavirus patients.
The US firm said its leronlimab treatment led to a greater improvement in patients after three days, which saw 90% of patient scores improve, than the placebo, which saw 71% of scores improve, in completed in Phase 2 trials.
CytoDyn chief executive Nader Pourhassan (pictured)said on Wednesday the drug showed “a statistically significant improvement” in treating the virus.
He added: “In the mild-to-moderate population, it is important to have a therapeutic option for COVID-19 in patients who are showing signs of rapid clinical deterioration.”
CytoDyn to register results in US, UK and EU
Pourhassan continued: “We will make a case for immediate approval of leronlimab for this population of COVID-19 patients, not only in the US, but in the UK and other countries around the world.”
The study also used The National Early Warning Score 2, a scale developed by Britain’s Royal College of Physicians to identify patients at risk for rapid clinical deterioration requiring critical care.
By this measure, patients in the leronlimab group were more than twice as likely to experience an improvement than the placebo group after two weeks. Half of leronlimab patients improved by that metric, compared to 20% of placebo patients.
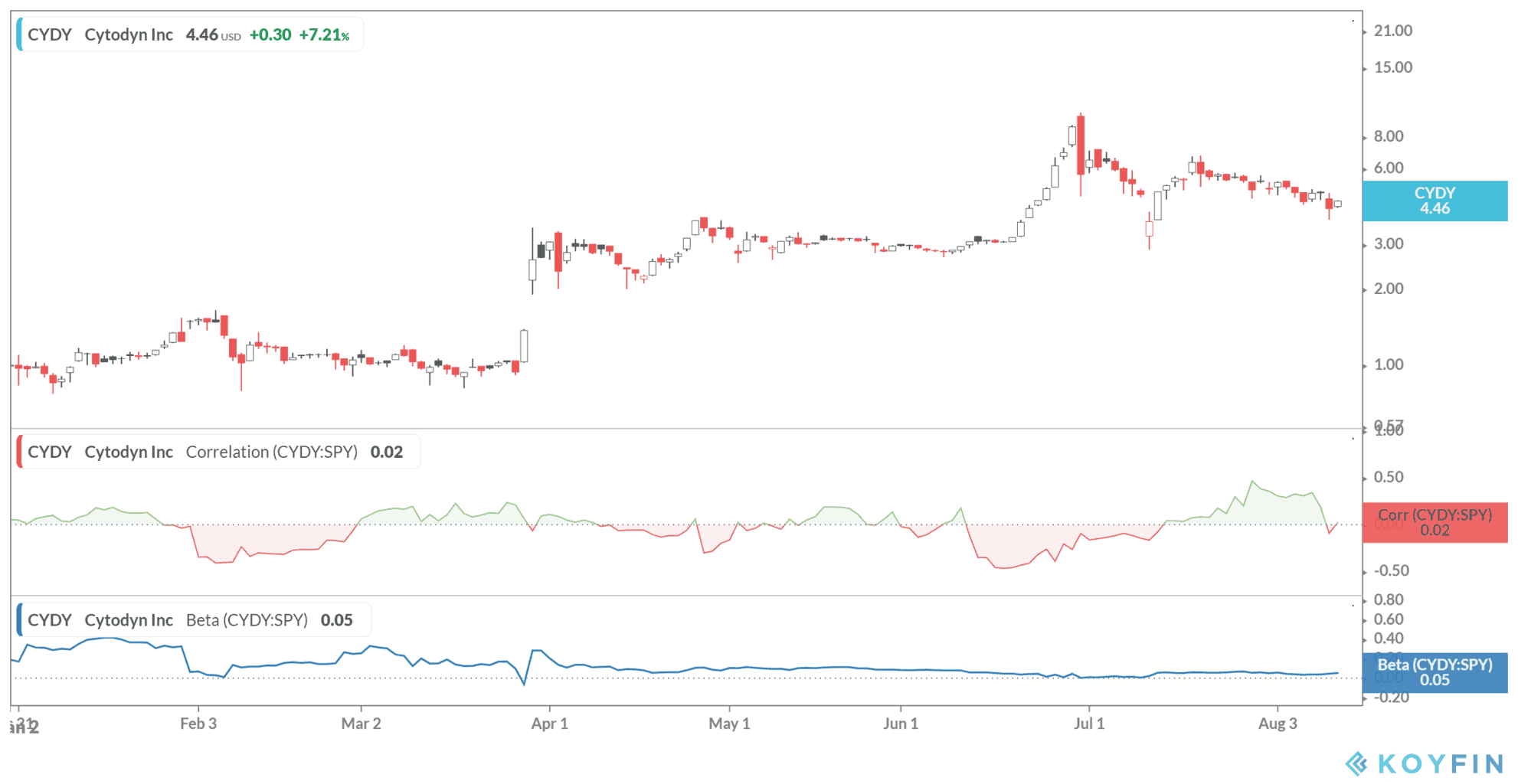
The Washington-based health firm, which also develops treatments for cancer and HIV, rose more than 7% in stock trading to close at $4.46 on Wednesday.
Biotech reaction
CytoDyn has a 14-day relative strength index (RSI) of 44.85 suggests the company is trading in technically neutral territory. RSI scores range from 0 to 100, where the stock is considered overbought when the index is above 70 and oversold when below 30.
CytoDyn, founded in 2002, said it will report its findings to the US Food and Drug Administration (FDA), the UK Medicines & Healthcare products Regulatory Agency (MHRA) and the EU European Medicines Agency.
CytoDyn is one of a host of firms looking for a treatment for coronavirus pandemic, which has soared to 20.7 million cases and killed 749,028 around the world, according to data from Johns Hopkins University.
Several other marketed drugs such as Novartis inhibitor Jakafi; AstraZeneca’s Calquence, and Amgen’s Otezla, among others, are being tested to see if they can treat respiratory complications associated with coronavirus.
You can check out a list of recommended stock brokers if you want to invest in stocks.
Trusted & Regulated Stock & CFD Brokers
What we like
- 0% Fees on Stocks
- 5000+ Stocks, ETFs and other Markets
- Accepts Paypal Deposits
Min Deposit
$200
Charge per Trade
Zero Commission on real stocks
64 traders signed up today
Visit Now67% of retail investor accounts lose money when trading CFDs with this provider. You should consider whether you can afford to take the high risk of losing your money.
Available Assets
- Total Number of Stocks & Shares5000+
- US Stocks
- German Stocks
- UK Stocks
- European
- ETF Stocks
- IPO
- Funds
- Bonds
- Options
- Futures
- CFDs
- Crypto
Charge per Trade
- FTSE 100 Zero Commission
- NASDAQ Zero Commission
- DAX Zero Commission
- Facebook Zero Commission
- Alphabet Zero Commission
- Tesla Zero Commission
- Apple Zero Commission
- Microsoft Zero Commission
Deposit Method
- Wire Transfer
- Credit Cards
- Bank Account
- Paypall
- Skrill
- Neteller
What we like
- Sign up today and get $5 free
- Fractals Available
- Paypal Available
Min Deposit
$0
Charge per Trade
$1 to $9 PCM
Visit Now
Investing in financial markets carries risk, you have the potential to lose your total investment.
Available Assets
- Total Number of Shares999
- US Stocks
- German Stocks
- UK Stocks
- European Stocks
- EFTs
- IPOs
- Funds
- Bonds
- Options
- Futures
- CFDs
- Crypto
Charge per Trade
- FTSE 100 $1 - $9 per month
- NASDAQ $1 - $9 per month
- DAX $1 - $9 per month
- Facebook $1 - $9 per month
- Alphabet $1 - $9 per month
- Telsa $1 - $9 per month
- Apple $1 - $9 per month
- Microsoft $1 - $9 per month
Deposit Method
- Wire Transfer
- Credit Cards
- Bank Account



